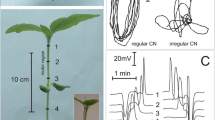
Abstract
Using apoplastic voltage- and ion selective microprobes, in barley leaves action potentials (APs) have been measured, which propagate acropetally as well as basipetally from leaf to leaf or from root to leaf following the application of mild salt stress (e.g. 30–50 mM KCl or NH4Cl) or amino acids (e.g. 1 mM glutamic acid or 5 mM GABA). Voltage changes were biphasic, followed an ‘all-or-none’ characteristic, and propagated at 20–30 cm min−1 irrespective of the direction. With the salt-induced APs, a strong initial depolarization is the main AP-releasing factor that first causes Ca2+ influx and then anion efflux. Ca2+ influx coincides with an initial slower depolarization, the rapid anion efflux causes the typical voltage ‘break-through’. Subsequently, K+-efflux starts after the depolarizing voltage has passed the K+ equilibrium potential (inversion of the K+ driving force). Glutamic acid and GABA induce APs not through membrane depolarization, but presumably by binding to a putative receptor or to ligand-gated Ca2+-conducting channels, respectively, followed by Ca2+ induced activation of anion efflux. APs are accompanied by transient apoplastic pH increase (about 1 unit), and by cytoplasmic pH decrease (about 0.5 units). The apoplastic pH change is interpreted as an indicator of stress, the cytoplasmic pH change as a prerequisite for defence related gene activation. Since APs are released by agents added in a moderate concentration range, it is suggested that they may serve as first and fast systemic signals following attack from pathogens.








Similar content being viewed by others
Abbreviations
- AP:
-
Action potential
- GABA:
-
γ-Aminobutyric acid
- Glu:
-
Glutamic acid
References
Abe T (1981) Chloride ion efflux during action potential in the main pulvinus of Mimosa pudica. Bot Mag 94:379–383
Alborn HT, Turlings TCJ, Jones TH, Stenhagen G, Loughrin JH, Tumlinson JH (1997) An elicitor of plant volatiles from beet armyworm oral secretion. Science 276:945–949
Beilby MJ (1984) Calcium in plant action potentials. Plant Cell Environ 7:415–421
Brenner ED, Stahlberg R, Mancuso S, Vivanco J, Baluska F, van Volkenburgh E (2006) Plant neurobiology: an integrated view of plant signaling. Trends Plant Sci 11:413–419
Cole KS (1968) Membrane, ions and impulses. University of California Press, Berkeley, pp 152–168
Cully DF, Vassilatis DK, Liu KK, Paress PS, Van der Ploeg LHT, Schaffer JM, Arena JP (1994) Cloning of an avermectin-sensitive glutamate-gated chloride channel from Caenorhabditis elegans. Nature 371:707–711
Davies E (1987) Action potentials as multifunctional signals in plants: a unifying hypothesis to explain apparently disparate wound responses. Plant Cell Environ 10:623–631
Davies E (2004) New functions for electrical signals in plants. New Phytol 161:607–610
Dennison KL, Spalding EP (2000) Glutamate-gated calcium fluxes in Arabidopsis. Plant Physiol 124:1511–1514
Dziubinska H, Trebacz K, Zawadzki T (2001) Transmission route for action potentials and variation potentials in Helianthus annuus L. J Plant Physiol 158:1167–1172
Favre P, Zawadzki T, Dziubinska A, Trebacz K, Greppin H, Degli Agosti R (1999) Repetitive action potentials induced in the liverwort Conocephalum conicum. Arch Sci 52:187–198
Favre P, Greppin H, Agosti RD (2001) Repetitive action potentials induced inArabidopsis thaliana leaves by wounding and potassium chloride application. Plant Physiol Biochem 39:961–969
Felle HH (1981) A study of the current-voltage relationships of electrogenic active and passive membrane elements in Riccia fluitans. Biochim Biophys Acta 646:151–160
Felle HH, Kondorosi É, Kondorosi Á, Schultze M (1998) The role of ion fluxes in Nod factor signaling in Medicago sativa. Plant J 13:455–463
Felle HH, Hanstein S, Steinmeyer R, Hedrich R (2000) Dynamics of ionic-activities in the apoplast of the sub-stomatal cavity of intact Vicia faba leaves during stomatal closure evoked by ABA and darkness. Plant J 24:297–304
Felle HH, Herrmann A, Hanstein S, Hückelhoven R, Kogel K-H (2004) Apoplastic pH-signalling in barley leaves attacked by the powdery mildew fungus (Blumeria graminis f.sp. hordei). Mol Plant Microbe Interact 17:118–123
Felle HH, Herrmann A, Hückelhoven R, Kogel (2005) Root-to-shoot signalling: apoplastic alkalinization, a general stress signal and defence factor in barley (Hordeum vulgare). Protoplasma 227:17–24
Fisahn J, Herde O, Willmitzer L, Peña-Cortez H (2004) Analysis of the transient increase in cytosolic Ca2+ during the action potential of higher plants with high temporal resolution: requirement of Ca2+ transients for induction of jasmonic acid biosynthesis and PINII gene expression. Plant Cell Physiol 45:456–459
Forterre Y, Skotheim JM, Dumais J, Mahadevau L (2005) How the venus flytrap snaps. Nature 433:421–425
Fromm J, Bauer T (1994) Action potentials in maize sieve tubes change phloem translocation. J Exp Bot 45:463–469
Fromm J, Fei HM (1998) Electrical signalling and gas exchange in maize plants of drying soil. Plant Sci 132:203–213
Gradmann D, Mummert H (1980) Plant action potentials. In: Spanswick RM, Lucas WJ, Dainty J (eds) Plant membrane transport: current conceptual issues. Elsevier North-Holland Biomedical Press, Amsterdam, pp 333–337
Graham JS, Hall G, Pearce G, Uyan CA (1986) Regulation of synthesis of proteinase inhibitor-I and inhibitor-II messenger RNAs in leaves of wounded tomato plants. Planta 169:399–405
He DY, Yazaki Y, Nishizawa Y, Takai R, Yamada K, Sakano K, Shibuya N, Minami E (1998) Gene activation by cytoplasmic acidification in suspension-cultured rice cells in response to the elicitor, N-acetylchitoheptaose. Mol Plant Microbe Interact 11:1167–1174
Herde O, Peña-Cortes H, Willmitzer L, Fisahn J (1998) Time-resolved analysis of signals involved in systemic induction of Pin2 gene expression. Bot Acta 111:383–389
Hope AB, Walker NA (1975) Action potentials in charophyte cells. In: The physiology of giant algal cells. Cambridge University Press, Cambridge, pp 111–119
Julien JL, Desbiez M-O, De Jaegher G, Frachisse JM (1991) Characteristics of the wave of depolarization induced by wounding in Bidens pilosa L. J Exp Bot 42:131–137
Kikujama M, Tazawa M (1982) Transient increase of intracellular Ca2+ during excitation of tonoplast-free Chara cells. Protoplasma 117:62–67
Lam HM, Chiu J, Hsie MH, Meisel L, Oliveira IC, Shin M, Coruzzi G (1998) Glutamate-receptor genes in plants. Nature 396:125–126
Maffei M, Bossi S, Spiteller D, Mithöfer A, Boland W (2004) Effects of feeding Spodoptera littoralis on Lima bean leaves. I. Membrane potentials, intracellular calcium variations, oral secretions, and regurgitate components. Plant Physiol 134:1752–1762
Malone M (1992) Kinetics of wound-induced hydraulic signals and variation potentials in wheat seedlings. Planta 187:505–510
Malone M (1994) Wound-induced hydraulic signals and stimulus transmission in Mimosa pudica L. New Phytol 128:49–56
Malone M (1996) Rapid, long-distance signal transmission in higher plants. Adv Bot Res 22:163–228
Mancuso S (1999) Hydrolic and electrical transmission of wound-induced signals in Vitis vinifera. Aust J Plant Physiol 26:55–61
Meyerhoff O, Müller K, Rolfsema MRG, Latz A, Lacombe B, Hedrich R, Dietrich P, Becker D (2005) AtGLR3.4, a glutamate receptor channel-like gene is sensitive to touch and cold. Planta 222:418–427
Mishra NS, Mallick BN, Sopory SK (2001) Electrical signal from root to shoot in Sorghum bicolor: induction of leaf opening and evidence for fast extracellular propagation. Plant Sci 160:237–245
Mithöfer A, Wanner G, Boland W (2005) Effects of feeding Spodoptera littoralis on Lima bean leaves. II. Continuous mechanical wounding resembling insect feeding is sufficient to elicit herbivory-related volatile emission. Plant Physiol 137:1160–1168
Nürnberger T, Nennstiel D, Jabs T, Sacks WR, Hahlbrock K, Scheel D (1994) High affinity binding of a fungal oligopeptide elicitor to parsley plasma membranes triggers multiple defence responses. Cell 78:449–460
Oostendorp M, Kunz W, Dietrich B, Staub T (2001) Induced resistance in plants by chemicals. Eur J Plant Pathol 107:19–28
Pickard BG (1973) Action potentials in higher plants. Bot Rev 39:172–201
Plieth C, Sattelmacher B, Hansen UP, Thiel G (1998) The action potential in Chara: Ca2+ release from internal stores visualized by Mn2+-induced quenching of fura-dextran. Plant J 13:167–175
Rhodes JD, Thain JF, Wildon DC (1996) The pathway for systemic electrical signal conduction in the wounded tomato plant. Planta 200:50–57
Rhodes JD, Thain JF, Wildon DC (1997) Evidence for physically distinct systemic signalling pathways in the wounded tomato plant. Ann Bot 84:109–116
Roblin G (1985) Analysis of the variation potential induced by wounding in plants. Plant Cell Physiol 26:255–261
Samejima M, Sibaoka T (1980) Changes in the extracellular ion concentration in the main pulvinus of Mimosa pudica during rapid movement and recovery. Plant Cell Physiol 21:467–479
Schwartz-Bloom RD, Sah R (2001) γ-Aminobutyric acid neurotransmission and cerebral ischemia. J Neurochem 77:353–371
Shepard VA, Beilby MJ, Shimmen T (2002) Mechanosensory ion channels in charophyte cells: the response to touch and salinity stress. Eur Biophys J 31:341–355
Stankovic B, Davies E (1997) Intercellular communication in plants: electrical stimulation of proteinase inhibitor gene expression in tomato. Planta 202:402–406
Stankovic B, Witters DL, Zawadzki T, Davies E (1998) Action potentials and variation potentials in sunflower: an analysis of their relationships and distinguishing characteristics. Physiol Plant 103:51–58
Steigner W, Köhler K, Simonis W, Urbach W (1987) Transient cytoplasmic pH changes in correlation with opening of potassium channels in Eremosphaera. J Exp Bot 39:23–36
Trebacz K, Stolarz M, Dziubinska H, Zawadzki T (1997a) Electrical control of plant development. In: Greppin H, Pennel C, Simon P (eds) Travelling shot on plant development. University of Geneva, Geneva pp 163–179
Trebacz K, Simonis W, Schönknecht G (1997b) Effects of anion channel inhibitors on light-induced potential changes in the liverwort Conocephalum conicum. Plant Cell Physiol 38:550–557
Walling LL (2000) The myriad plant responses to herbivores. J Plant Growth Regul 19:195–216
Wayne R (1994) The excitability of plant cells: with special emphasis on characean internodial cells. Bot Rev 60:265–367
Wildon DC, Thain JF, Minchin PEH, Gubb IR, Reilly AJ, Skipper YD, Doherty HM, O’Donnell PJ, Bowles DJ (1992) Electrical signalling and systemic proteinase inhibitor induction in the wounded plant. Nature 360:62–65
Wilkinson S (1999) pH as a stress signal. Plant Growth Regul 29:87–99
Williamson RE, Ashley CC (1982) Free Ca2+ and cytoplasmic streaming in the alga Chara. Nature 296:647–651
Zawadzki T, Dziubinska H, Davies E (1995) Characteristics of action potentials generated spontaneously in Helianthus annuus. Physiol Plant 93:291–297
Acknowledgments
The financial support by the Deutsche Forschungsgemeinschaft (Fe 213/15-1) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Felle, H.H., Zimmermann, M.R. Systemic signalling in barley through action potentials. Planta 226, 203–214 (2007). https://doi.org/10.1007/s00425-006-0458-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-006-0458-y